Blog
Elevating Patient Safety: Rethinking Complaint Management in MedTech
Navigation
NavigationIntroduction
In the realm of medical devices and innovative treatments technological advancements are transforming what was once considered impossible into reality.
While these innovations significantly improve patient outcomes, they also introduce substantial risks associated with patient safety that must be carefully controlled. To mitigate such risks, manufacturers must continuously monitor device performance in real-world use and leverage patient and user data to strengthen product safety profiles.
Customer complaints play a critical role in this process. They serve as early indicators of potential safety or performance issues and provide actionable insights into how devices perform outside controlled pre-market environments. Many latent risks emerge after commercialization, making complaint management an indispensable tool for quality assurance and post-market surveillance.
Effective complaint management in the medical device industry is therefore not merely an operational or regulatory obligation; it is foundational to regulatory compliance, patient safety, product quality, and brand credibility. When executed well, it supports proactive risk management and continuous product improvement. This article explores the medical device complaint lifecycle, examines the inherent challenges in maintaining effective complaint management systems, and outlines practical, field-tested solutions aligned with global regulatory expectations and real-world operational challenges.
Our focus is on delivering MedTech Complaint Management Services that strengthen governance, consistency, and speed while maintaining strict compliance.
Overview of the Medical Device Complaints Lifecycle
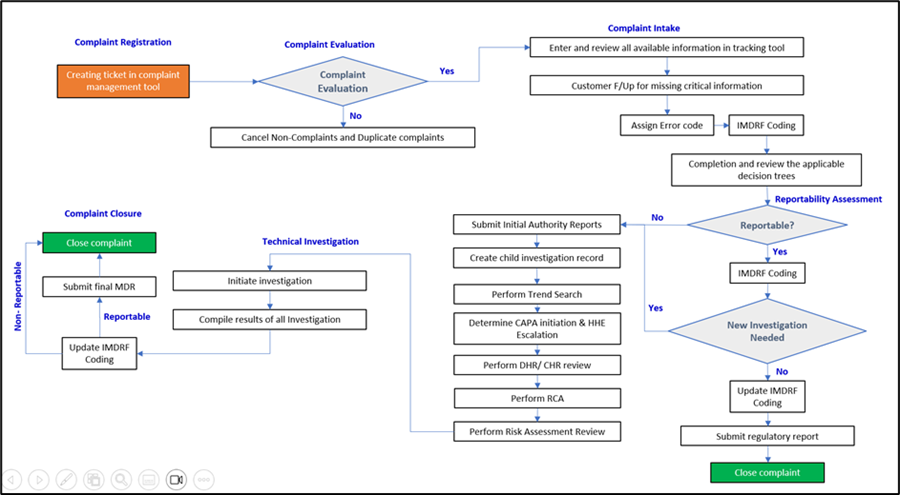
The medical device complaint lifecycle typically begins when a customer experiences an issue during device use. Initial contact may occur through a Field Safety Engineer (FSE), or Service Executive, during which the concern is documented and reported to the Designated Complaint Centre of the manufacturer.
Each manufacturer is mandated by regulation to establish a complaint handling unit, often referred to as a Designated Complaint Unit (DCU) or a Designated Complaint Centre (DCC). The Designated Complaint Unit then evaluates the information, determines whether the issue meets the definition of a complaint, and assesses its reportability in accordance with applicable regulatory requirements. For reportable events, the complaint unit prepares and submits mandatory reports to regulatory authorities within prescribed timelines.
If the product is returned, a technical investigation is initiated to confirm defects, assess device performance, and identify root causes. Investigation findings are incorporated into the final complaint record and may trigger corrective and preventive actions (CAPA), risk management updates, or design and process improvements. This structured lifecycle enables the resolution of individual complaints while strengthening post-market surveillance and driving continuous improvement across the product lifecycle.
Despite this defined framework, many organizations struggle to maintain consistency, speed, and compliance while managing complaint volumes at scale amid complex product ecosystems and evolving global regulations.
Below is an illustrative flowchart:
Organizations often rely on post-market surveillance and complaint reporting services to ensure timely submissions and consistent feedback loops aligned with evolving regulations (e.g., MDR, FDA).
Complexities and Challenges in Maintaining an Effective Complaint Management System
Medical device complaint management is inherently complex due to regulatory rigor, diverse product portfolios, global operations, and fragmented data environments. These challenges are further intensified by cross-functional dependencies and increased regulatory scrutiny through post-market surveillance programs.
- Inconsistent Procedures and Definitions
In many organizations, complaint handling procedures differ across legal entities, product types, and divisions. The absence of globally harmonized processes results in inconsistent intake criteria and unclear definitions of what constitutes a complaint. These inconsistencies often lead to incorrect prioritization, incomplete documentation, overdue complaint records, and delayed reportability determinations.
Unclear guidelines also contribute to misclassification between service requests, non-reportable complaints, and serious events such as injuries or device malfunctions. This creates regulatory risk, particularly when reportable events are not identified or escalated in a timely manner, leading to duplication of effort and time in an already burdened system.
- Manual, Reactive, and Inefficient Processes
Manual processes continue to dominate complaint handling in many organizations, leaving significant room for human error and inconsistent decision-making. This makes it difficult to track record progress, good-faith effort, perform timely trend analysis, and ensure consistent application of regulatory requirements which can have serious regulatory implications. As a result, complaint cycle time increases, particularly for reportable cases where multiple undue reports are often generated.
Complaint management often remains reactive rather than proactive, with limited preventive controls and ineffective quality checks. This reactive stance frequently leads to non-conformities identified during regulatory audits, and the subsequent remediation activities add further strain to already overburdened systems.
- Investigation and Data Management Gaps
Complaint investigations are frequently dependent on multiple internal and external stakeholders, such as service providers or distributors. Insufficient follow-up attempts to gather missing information or retrieve returned products often result in investigations that focus on symptom identification rather than true root cause analysis.
Inadequate tracking of product identifiers, such as lot numbers or serial numbers, further complicates investigations. Fragmented systems and poor data integrity increase the risk of missed timelines, delayed complaint closures, and weak feedback loops to design, manufacturing, and risk management teams. Over time, these gaps limit the organization’s ability to detect systemic issues and implement effective corrective actions.
- Limited Effectiveness Metrics
Most organizations measure complaint management effectiveness primarily through regulatory submission compliance and complaint closure timelines. While these metrics are essential, they provide limited insight into quality trends, root cause, and safety signal detection. Without broader performance indicators, opportunities to improve device safety, reliability, and performance are often missed.
Ultimately, the goal is to improve patient safety through effective complaint resolution systems, which require harmonized processes, reliable data, and clear accountability.
Practical Solutions to Improve Complaint Handling Efficiency and Effectiveness
Addressing the complexities of medical device complaint management requires a strategic, technology-enabled, and process-driven approach.
- Process Standardization and Governance
Organizations should implement globally standardized Complaint handling procedures across legal entities, product types, and divisions to eliminate variability. Clear, well-documented guidelines supported by robust training programs to ensure accurate intake, proper triage, and timely reportability assessments. The use of quick reference tools for critical steps can further improve usability and reduce processing time.
Well-designed governance frameworks are the backbone of end-to-end MedTech complaint management solutions for compliance, aligning local operations to global standards.
- Intelligent Intake and Automation
To reduce ambiguity and misclassification, organizations should adopt intelligent complaint intake systems powered by artificial intelligence (AI) and natural language processing (NLP). These systems can automatically categorize complaints, assess reportability, and flag high-risk cases, ensuring consistency and speed. Automation can also streamline investigations by integrating workflows across quality, regulatory, R&D, and field service teams, reducing dependency on manual follow-ups, and minimizing delays.
A critical enabler here is AI-driven complaint analytics for medical devices, which accelerates triage and improves coding accuracy while reducing manual error.
- Centralized Systems and Real-Time Visibility
Implementing centralized complaint management platforms with real-time dashboards enables effective tracking of good-faith effort activities, product return status, investigation progress, and closure timelines. Enhanced visibility improves accountability, transparency, and regulatory readiness. Advanced analytics and trend monitoring tools further strengthen feedback loops by identifying recurring issues, root cause patterns, and emerging safety signals.
For global rollouts, organizations often partner with global medical device complaints handling consulting to tailor templates, taxonomies, and workflows across regions.
- Digitization and Proactive Quality Controls
Digitizing complaint handling processes through electronic records, automated reminders, and escalation triggers significantly reduce human error and shortens cycle times. Proactive quality controls, such as automated audit-readiness checks and integrated CAPA systems, help prevent non-conformities and reduce the burden of regulatory remediation.
What are the benefits of automated complaint management in MedTech? Faster cycle times, better data integrity, and consistent regulatory outputs across markets.
- Predictive and Performance-Driven Management
Shifting from reactive to predictive complaint management through AI-driven risk modelling and early warning systems enhances patient safety and regulatory compliance. Effectiveness metrics should extend beyond submission timelines to include quality indicators such as root cause, trend analysis outcomes, recurrence rates, and signal detection performance. This broader measurement framework enables data-driven decision-making and supports continuous improvement.
Modern platforms provide scalable complaint management solutions to reduce MedTech compliance risks, enabling capacity growth without sacrificing controls.
You can optimize MedTech complaint investigation processes by adhering to a structured intake, required fields, and closed-loop evidence collection that feeds downstream CAPA and risk management.
Conclusion
As regulatory expectations around medical device complaint handling continue to intensify, with greater emphasis on effective feedback loops and demonstrable process performance, complaint management has become a strategic quality function rather than a reactive obligation. Regulators expect systems that not only capture complaints but also consistently analyze, escalate, and close them in a controlled and timely manner. To meet these expectations, medical device manufacturers must align their complaint handling frameworks with the intent of the regulations and design processes that are robust, transparent, and efficient.
A streamlined, linear complaint management lifecycle is foundational to this objective. Clear instructions, well-defined roles, and practical job aids must be established to guide all stakeholders involved in complaint intake, evaluation, investigation, and closure. Equally critical is the availability of accessible and user-friendly tools that support end-to-end complaint processing, minimize variability, and reduce the risk of delays or misinterpretation. Strong quality measures, along with tools that monitor complaint progress, record aging, and identify bottlenecks, are essential. Visual dashboards that provide real-time visibility into complaint volumes and stages, further enable proactive oversight and timely decision-making.
The effective use of technology is a key enabler in achieving consistency, compliance, and scalability in complaint management. Advanced technologies such as NLP and AI can support smart identification and categorization of complaints, automated or assisted coding, and decision-making, ensuring uniform interpretation and classification across the organization. This reduces dependency on individual judgment, minimizes the risk of reportability of misses due to human intervention, and enhances overall data quality. Furthermore, regulatory reporting requirements often differ across regions, leading to duplication of effort and potential inconsistencies when reports are prepared manually. Technology-enabled systems can leverage information already captured within a complaint record to automatically generate multiple regulatory report formats, eliminating redundant work while ensuring uniformity and accuracy of information across submissions.
By integrating clear processes, robust monitoring mechanisms, and intelligent, technology-driven solutions, medical device manufacturers can transform complaint management into a highly controlled, efficient, and value-driven system. Such an approach not only strengthens regulatory compliance but also improves data integrity, operational efficiency, and, most importantly, patient safety.
Sustained outcomes hinge on designing and implementing robust MedTech complaint tracking systems that connect investigations, CAPA, risk, and regulatory reporting end-to-end. When combined with post-market surveillance and complaint reporting services for MedTech, manufacturers can demonstrate control and audit readiness across jurisdictions (e.g., MDR and FDA 21 CFR Part 820).